The Chemistry Of LifeA. Key Forces Acting On Matter
b. Matter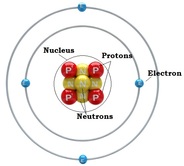 Atom Atom
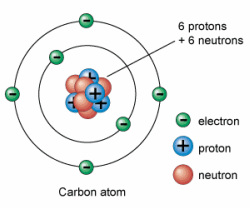
1. Atoms: a basic unit of matter with a specific number of protons for each element. Atoms have a zero charge when isolated. This means that the number of electrons will equal the number of protons in an atom.
Image: Source: Live Science> 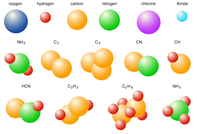 Molecules Molecules
2. Elements: consists of
all the same type of an atom. 3. Compounds: 2 or more elements in definite proportions. Image Source: Andrew's Blog> 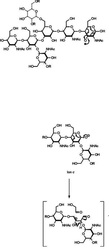
4. Molecules: 2 or more elements chemically bonded.
5. Ions: positively and negatively charged atoms. 6. Radioactivity: energy released when atomic nuclei are unstable due to radiation. 7. Isotopes: atoms of the same element that differ in the number of neutrons. Image Source: Science Direct> |
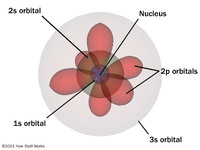
c. The AtomThe Quantum Mechanical Model of the atom is the current theory of the atom that states that the electrons have a vibrating wave character and uncertain positions. Sometimes,they are close to the nucleus and sometimes away from it.
D. Sub Atomic Particles 3 Sub Atomic Particles:
F. Water And Its Important to Life
|
E. Chemical Bonds
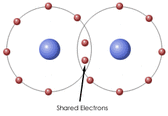
1. Covalent Bonds: a covalent bond is formed when electrons are shared between atoms.
Examples:
Image Source: The Encyclopedia of Science> 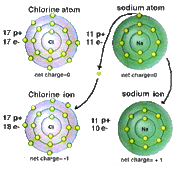
2. Ionic Bonds: an ionic bond is formed when one or more electrons are transferred from, one atom to another.
Examples:
Image Source: IB Chemistry> |
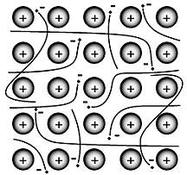
3. Metallic Bonds: metallic bonds are the electromagnetic interaction between lost electrons, called conduction electrons.
Examples:
Image Source: Kens Chemistry> 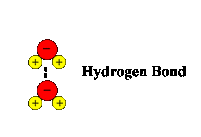
4. Hydrogen Bonds: hydrogen bonds are formed when a charged part of a molecule having polar covalent bonds forms an electrostatic interaction with a substance of opposite charge.
Examples:
|
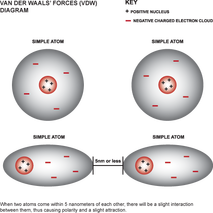
5. Van der Waals Forces: when molecules are hold together, a slight attraction can develop between the opposite charged regions of nearby molecules.
Examples:
Image Source: Zimie Science>
Examples:
- Hydrogen Bonds
- Dispersion Forces
- Dipole Interactions
Image Source: Zimie Science>