Enzyme-Controlled Reactions
A. ProblemThe purpose of this lab is to observe how enzymes act on substrates to produce products. We will observe how enzyme activity is altered when the enzyme environment's pH is changed.The purpose of this lab is to observe how enzymes act on substrates to produce products. We will observe how enzyme activity is altered when the enzyme environment's pH is changed.
B. Hypothesis
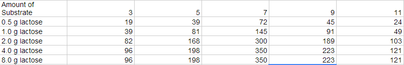
E. Results
In all the cases while I increased the levels of lactose, the levels of pH increased specially when I added 4 and 8 g of lactose. In addition, both (4 and 8g of lactose) gave me the same number of molecules (x10^6) formed per minute.
While I added more levels of lactose the number of molecules (x10^6) increased. |
C. Materials
D. Procedure
|
-Reaction Equation
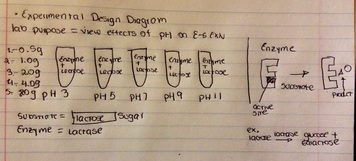
-EXPERIMENTAL design Diagram
F. cONCLUSION
Virtual Lab> Journal Questions
Conclusion:
The purpose of this experiment was to see how different amounts of lactose in pH levels affected the production of molecules (x10^6) formed per minute. In my hypothesis I stated that If I make the pH of the lactase reaction more acidic, the products formed would be a lot less than what is normally produced and if I make the pH more basic, the products formed would be less than what is normally produced. The experiment supported my hypothesis by showing that if I added lactase in basic pH, the number of molecules formed per minute (x10^6) decreases from its normal production and If I added lactose in acid pH levels the production of molecules decreased more than the basic because there are no more enzymes.
- The relationship between substrate concentration and the initial reaction rate of an enzyme-catalyzed reaction its a linear relationship. When the initial reaction rate as substrate concentration increases the number of molecules of product formed per minute (x10^6) increases. and there are not more enzymes initially.
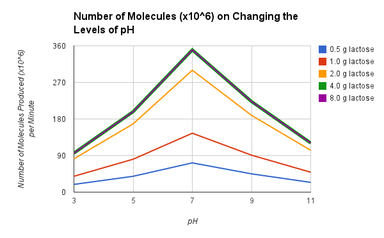
- The maximum initial reaction rate for the lactase enzyme at pH 7 is 350 molecules per minute (x10^6).
- The maximum initial reaction rate cannot be reached at low lactose concentrations because, the amount of lactose with the amount of pH cant reach the maximum initial react in rate because it has the higher rate.
- The optimum pH level is 7 because with all the different amount of substrates, pH 7 has always high molecules formed per minute.
- This suggests that the importance of temperature-regulating mechanisms in organisms change the reaction rate because it regulates mechanics in organisms.
- The producer of this product need to be concerned about the pH because it has to be exact (pH 7). Lactase likes a neutral environment
Conclusion:
The purpose of this experiment was to see how different amounts of lactose in pH levels affected the production of molecules (x10^6) formed per minute. In my hypothesis I stated that If I make the pH of the lactase reaction more acidic, the products formed would be a lot less than what is normally produced and if I make the pH more basic, the products formed would be less than what is normally produced. The experiment supported my hypothesis by showing that if I added lactase in basic pH, the number of molecules formed per minute (x10^6) decreases from its normal production and If I added lactose in acid pH levels the production of molecules decreased more than the basic because there are no more enzymes.